
Hamed Abdollahi
Molecular Biologist
Genomics Postdoctoral Fellow
DVM, PhD
Confidently, I Can Help Solve Your Lab’s Most Critical Challenges!
Welcome to the forefront of molecular biology innovation. As an experienced molecular biologist, I'm here to assist you through the complexities of laboratory research and help you overcome your most pressing challenges.
QUALITY MANAGEMENT SYSTEMS
Accurate and reliable laboratory results are crucial for R&D, diagnosis, surveillance, and trade. These results are guaranteed through the implementation of robust management systems that ensure consistent testing and calibration methods. With specialized training in laboratory management systems, quality control, and technical operations under ISO/IEC 17025 standards, I ensure that all lab processes achieve the highest levels of accuracy, reliability, and consistency. Laboratory quality management encompasses not only the technical aspects but also the managerial and operational components necessary for precise testing, interpretation, and reporting.
Biological Risk Assessment
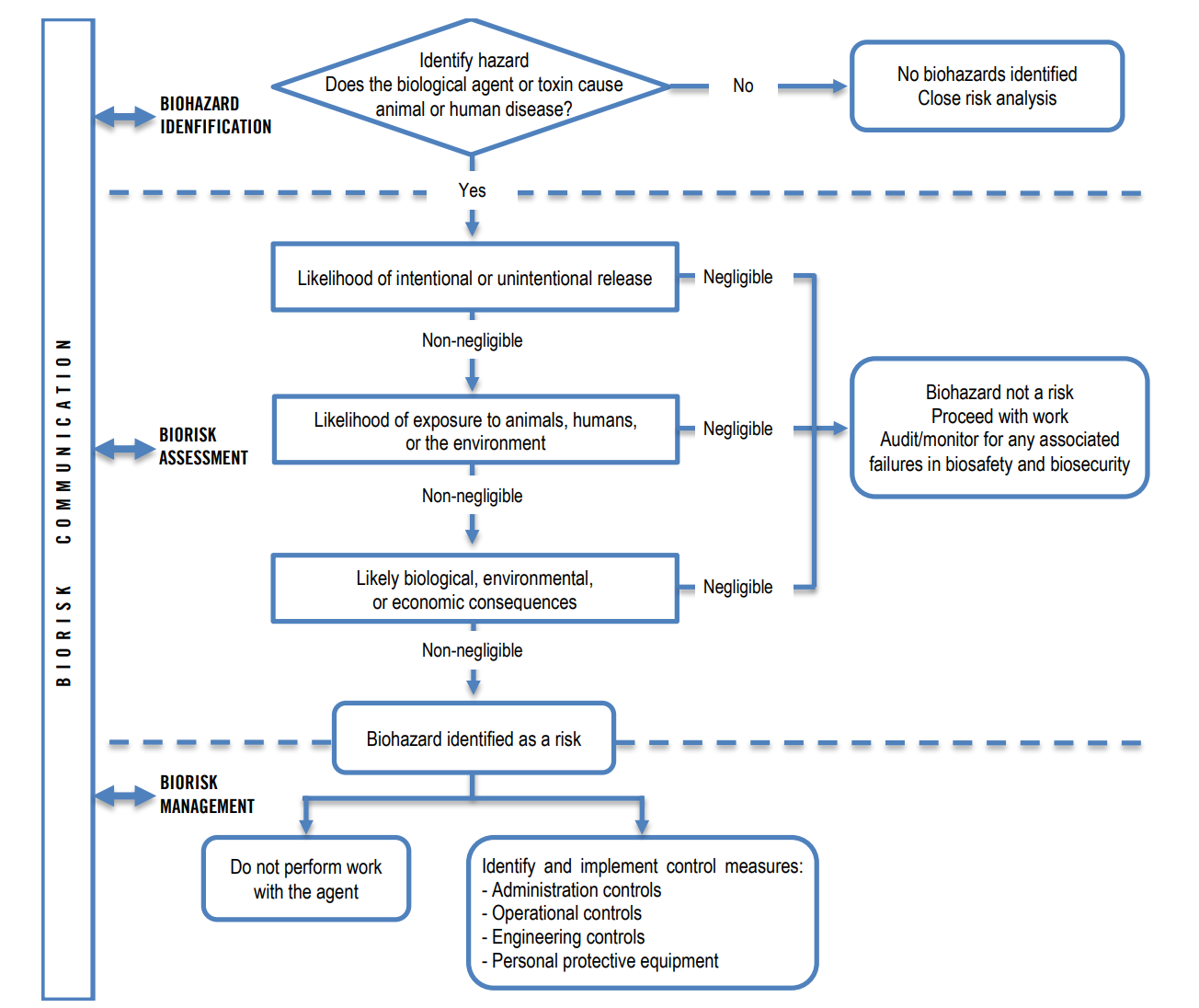
To handle the complexity of risk management in a molecular biology lab, a biological risk management advisor with extensive knowledge and experience is indeed crucial. This professional plays a vital role in evaluating and mitigating risks associated with biohazardous materials and procedures. Here's how such an advisor can assist in the biological risk analysis process:
1- Comprehensive knowledge of biohazards, organisms, and diseases.
2- In-depth understanding of molecular biology techniques and their associated risks.
3- Experience in evaluating laboratory facilities, protocols, and procedures.
By leveraging the expertise of a biological risk management advisor, molecular biology labs can effectively navigate the complexities of risk assessment and management, ensuring the safety of personnel, the community, and the environment while facilitating cutting-edge research.
Diagnostic ASSAY DEVELOPMENT AND VALIDATION
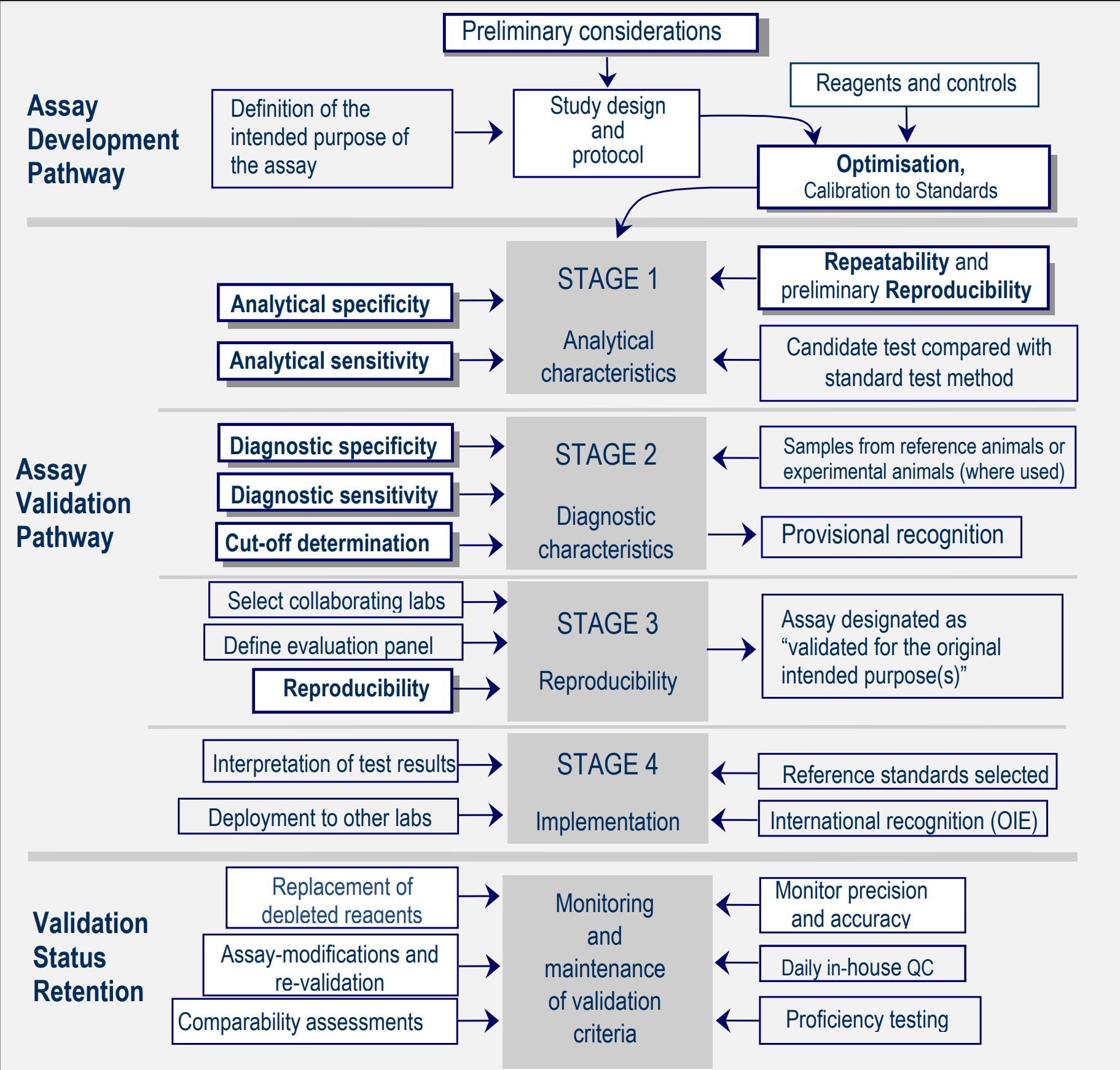
I offer a complete suite of molecular lab services, acting as a catalyst for the development of advanced diagnostics and testing kits. My expertise lies in evaluating and validating kit results, investigating the latest innovations in molecular diagnostics, and driving research and development to push the field forward. Drawing from extensive experience, I understand the critical importance of every step in the diagnostic process, from the selection, collection, preparation, and preservation of samples, to their management. I ensure precision at each phase, focusing on: Defining the intended purpose(s) Optimisation and standardisation Ensuring repeatability, analytical sensitivity, and specificity Setting diagnostic thresholds (cut-offs) Achieving high diagnostic sensitivity and specificity My goal is to foster innovation and deliver impactful, reliable solutions that drive progress in molecular diagnostics.
COLLECTION, SUBMISSION AND STORAGE OF DIAGNOSTIC SPECIMENS

Drawing from extensive experience in molecular laboratory techniques, I provide a complete laboratory package that encompasses everything from the fundamental principles of Good Laboratory Practice (GLP) to the most advanced sequencing and spectrometry methods. My expertise ensures that every step of the process meets the highest standards of precision and accuracy, offering unparalleled support for research and development needs.
| Protocol | Laboratory | Download |
|---|---|---|
| Guidance document for WGS-laboratory procedures | EU | SOP |
| Detection of Lyssavirus by the Rabies Tissue Culture Infection Test (RTCIT) | ANSES | SOP RTCIT |
| Guidance for genomic monitoring of Avian Influenza Virus (AIV) | IZSVe | |
| Preparation and processing of swab pools for detection of Influenza A Virus | APHA | |
| Isolation and characterization of Avian influenza viruses using SPF embryonated chicken eggs and haemagglutination inhibition test (Released on 25/01/24) | IZSVe | SOP VIR 005 |
| Isolation and characterization of Newcastle disease viruses using SPF embryonated chicken eggs and haemagglutination inhibition test (Released on 12/10/21) | IZSVe | SOP VIR 007 |
| Influenza – Neuraminidase Inhibition Test | APHA | |
| Detection of antibodies to type A influenza virus by agar gel immunodiffusion assay (AGID) (Released on 23/02/23) | IZSVe | SOP IMM 063 |
| Detection of subtype-specific antibodies to Avian influenza virus by haemagglutination inhibition test (Released on 29/12/20) | IZSVe | SOP IMM 064 |
My expertise extends across the entire PCR and real-time PCR workflow—from developing, evaluating, and improving various genome extraction procedures to performing complex multiplications, including reverse transcriptase applications, and concluding with diverse electrophoresis techniques.Following PCR, my skills extend to sequencing, where I began with Sanger sequencing to address research needs for zoonotic disease agents such as Influenza virus and Salmonella.


My expertise extends across the entire PCR and real-time PCR workflow—from developing, evaluating, and improving various genome extraction procedures to performing complex multiplications, including reverse transcriptase applications, and concluding with diverse electrophoresis techniques.Following PCR, my skills extend to sequencing, where I began with Sanger sequencing to address research needs for zoonotic disease agents such as Influenza virus and Salmonella.
| Protocol | Laboratory | Download |
|---|---|---|
| Detection of antibodies to Newcastle disease virus by haemagglutination inhibition test (Released on 30/12/20) | IZSVe | SOP IMM 065 |
| Sample preparation and nucleic acids isolation for the detection and typing of Avian influenza virus and Avian Orthoavulavirus type 1 by molecular methods (Released on 28/09/23) | IZSVe | SOP VIR 1000 |
| Detection of type A Avian influenza virus by real-time RT-PCR (Heine et al., 2015; Laconi et al., 2020) (Released on 28/09/23) | IZSVe | SOP VIR 018 |
| Detection of type A influenza virus by real-time RT-PCR (Nagy et al., 2021) (Released on 07/04/21) | IZSVe | SOP VIR 1003 |
| Detection of Eurasian H5 Avian influenza virus by real-time RT-PCR (Slomka et al., 2007) (Released on 09/04/21) | IZSVe | SOP VIR 143 |
| Detection of Eurasian H5 Avian influenza virus by One-step RT-PCR and Sanger sequencing of the hemagglutinin cleavage site (Slomka et al., 2007) (Released on 29/09/23) | IZSVe | SOP VIR 125 |
| Detection of Eurasian H7 Avian influenza virus by real-time RT-PCR (Slomka et al., 2009) (Released on 09/04/21) | IZSVe | SOP VIR 144 |
| Detection of Eurasian H7 Avian influenza virus by real-time RT-PCR (Van Borm et al., 2010 modified) (Released on 07/04/21) | IZSVe | SOP VIR 1001 |

Over the years, I have honed my ability to purify viral agents and create master species through cell culture techniques and virological processes. This expertise is complemented by extensive experience in serological assays like ELISA, where I evaluate and compare the functionality of ELISA kits. The use of them during certifying and evaluating the real status of specific antibody free (SAF) and specific pathogen free(SPF) animals were a big acheivement for me.


I have also utilized mass spectrometry and high-performance liquid chromatography (HPLC) to detect microcystins and profile pathogen proteins, covering the entire process from sampling to data analysis and interpretation with unmatched precision. My work adheres to the highest standards, including those set by FAO, WOAH, ISO, and GLP principles, within biosafety level 2, 2+, and 3 laboratories. The specialized practices, instruments, and facilities associated with these standards have become routine in my daily operations.
| Protocol | Laboratory | Download |
|---|---|---|
| Detection of Eurasian H7 Avian influenza virus by One-step RT-PCR and Sanger sequencing of the hemagglutinin cleavage site (Slomka et al., 2007) (Released on 29/09/23) | IZSVe | SOP VIR 126 |
| Detection of H9 Avian influenza virus by real-time RT-PCR (Panzarin et al., 2022) (Released on 17/06/22) | IZSVe | SOP VIR 014 |
| HA and NA subtyping of Avian influenza virus by real-time RT-PCR (Hassan et al., 2022; James et al., 2018; Hoffmann et al., 2016) (Released on 28/09/23) | IZSVe | SOP VIR 1004 |
| Avian Influenza virus (AIV) typing by reverse transcription PCR using a universal primer set (Released on 06/19) | IZSVe | |
| Detection of Avian Orthoavulavius type 1 (AOAV-1) by real-time RT-PCR (Sutton et al., 2019) (Released on 28/09/23) | IZSVe | SOP VIR 151 |
| Detection and pathotyping of Avian Orthoavulavirus type 1 (AOAV-1) by one-step RT-PCR and Sanger sequencing of the fusion protein cleavage site (De Battisti et al., 2013) (Released on 09/04/21) | IZSVe | SOP VIR 063 |
| Detection and pathotyping of Avian Orthoavulavirus type 1 (AOAV-1) by one-step RT-PCR and Sanger sequencing of the fusion protein cleavage site (Kant et al., 1997) (Released on 29/09/23) | IZSVe | SOP VIR 1002 |
| Laboratory standard operating procedure for MLVA of Salmonella enterica serotype Typhimurium | ECDC | SOP 1109 |
| Brucellosis EURL Specifications for validation of antigen for Rose Bengal Test | ANSES | SOP |
| Standard Operating Procedure Brucella real time PCR | ANSES | SOP |
This comprehensive skill set positions me as a valuable asset in the field of vaccine and drug discovery, where meticulous laboratory practices, advanced techniques, and strict adherence to international standards are paramount.
